Wide World of Vaccines
November 2021
Confusing Messages on Antibody Titers, the Delta-Plus Variant, and What’s with Monoclonal Antibodies?

Column Editor: Christopher Harrison, MD | Professor of Pediatrics, UMKC School of Medicine | Clinical Professor of Pediatrics, University of Kansas School of Medicine
With more children becoming candidates for a COVID-19 vaccine, how do we interpret data that continually flows from myriad sources, e.g., the internet, published articles, and our friends and colleagues? Every day or so, it seems, a new study or report indicates that one or another vaccine induces fewer or more antibodies, or provides less or more protection, or is better or worse against the Delta variant. Most recently, we’re hearing about the newest variant to catch the attention of the press: Delta-plus.
Confusing vaccine efficacy reports and more breakthrough infections.
1. Protection against what? It can be challenging to understand this critical question about what antibody titers mean. The goal of vaccines is to prevent disease. In an ideal world, vaccines would prevent any form of infection. But preventing a local infection of the mucosa, for instance in the nasal passages, can be difficult. When the pathogen infects through the mucosa, antibodies induced by injectable vaccines exist mostly in serum, not at the point of attack, the mucosa. Only about 10% of serum antibodies reaches the mucosa. So even a vaccine that is highly effective at preventing systemic or generalized respiratory disease could allow surface mucosal infection (and therefore a positive PCR test result).
2. Which immune system component provides protection? Functional antibodies (i.e., those that are protective) are only part of the total antibody products the body makes after infection or vaccine. Most commercial tests measure both functional and non-functional antibodies in the hope that the total is a surrogate for neutralizing functional antibodies that we want to identify. Neutralizing antibodies are tested primarily in research labs and during studies. But even “neuts,” as they are called, are NOT directly comparable from one lab to another. Also remember that we still are not sure what constitutes a protective neut titer.
3. Is a high titer required for protection? Yes, until memory-dependent immune responses mature. In the first three to six months post vaccine, circulating antibodies from the initial vaccine series are plentiful, but then begin to wane. Thus, disease protection is due to the high titer of the initial batch of circulating antibodies. But how much protection remains after the first batch of circulating antibody starts to wane, as it does six to 12 months after most vaccines (and post most infections)? At this point, immune memory (memory T-cells and memory B-cells) is crucial. When mucosal re-exposure occurs many months post vaccine, and the virus begins to enter cells in order to replicate (this initial replication is detectable by PCR), immune memory cells recognize the invasion. These memory cells mobilize, make copies of themselves, and produce another antibody batch (usually requiring three to seven days), shutting down further virus spread before it leads to disease. The second antibody batch is usually larger and more potent. The result is a “breakthrough infection” with no or minimal symptoms with only a few days of viral shedding, even after vaccines that are highly protective against more-than-minimal disease and death. Most breakthrough infections are mild or asymptomatic, except for people with multiple underlying conditions or compromised immune systems.
Does needing a booster mean the vaccine doesn’t work?
All three vaccines approved for emergency use authorization (EUA) dramatically lower a person’s chances of hospitalization or death. The booster dose tricks the immune memory cells into thinking SARS-CoV-2 is invading so that they mobilize and create another batch of antibodies. This approach is the same one we currently use for most vaccines. For example, we know tetanus vaccines are very effective. But to maintain effectiveness, we need three to four tetanus vaccine doses in childhood to establish strong immune memory, and then a booster every 10 years. So, we should not be surprised if evidence shows that we will need COVID-19 boosters at intervals, and possibly variant-specific boosters.
Delta-plus
We are familiar with the Delta variant arising in India early this year. But a sub-variant, Delta-plus, with newer mutations also arose in India in June 2021. Data are accumulating that Delta-plus is spreading in many countries. This variation appears to be similar in severity to the original Delta virus but is about 10-15% more contagious. Only time will tell if it supplants Delta, which now comprises >90% of U.S. infections. The good news is that current vaccines and their antibodies are as protective against Delta-plus as against Delta itself. At this point, then, new vaccines don’t seem necessary.
Even so, we need to recognize that antibodies from natural infections due to non-Delta viruses are not necessarily sufficient for protection.1 Hence the need for boosters six to nine months post current vaccines, with the exception of the Johnson & Johnson vaccine, which requires boosters two months post the single dose. We also need to know that Delta and Delta-plus are resistant to monoclonal antibodies (like the cocktail former president Trump got). For example, Delta is resistant to bamlanivimab, while Delta-plus is resistant to bamlanivimab and etesevimab.2 (See Table for current monoclonal antibody formulations and their activity against the original and variant viruses.3,4)
Complicating our efforts at an effective response to these unfolding challenges are the ongoing assaults on scientific evidence. For instance, a Florida school had been requiring teachers, staff and children who had received COVID-19 vaccine to quarantine for 30 days and not come to school. It went further to recommend that anyone with plans to get vaccinated hold off until summer. With no supporting evidence, the school claimed that vaccinated people are “shedding” and contagious for 30 days. Recently, the school relented once the Department of Education declared that the policy jeopardizes the school’s federal funding. Guess what? The school rescinded its policy.5 Ah, let’s enjoy the little victories for truth.
Table. In vitro neutralizing activity of monoclonal antibodies (MoAb) for potential clinical use against SARS-CoV-2. See footnotes for manufacturer and product generic names.
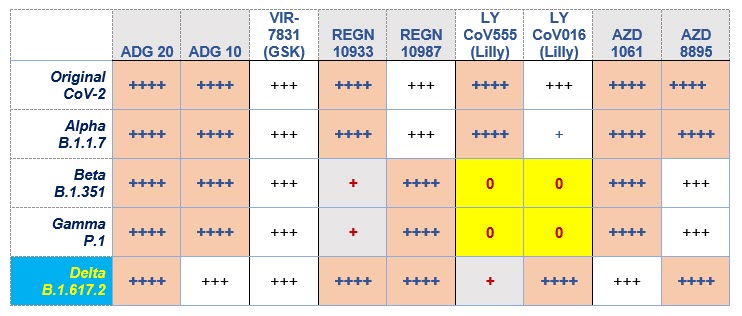
Key for IC50 (50% inhibitory concentration [neutralization activity]) concentration in mcg/mL: Best activity is when less monoclonal antibody (MoAb) is able to neutralize any given virus, so lower antibody concentration values mean more potency.
0 = >10 mcg/mL, + = 1-10 mcg/mL, ++ = 0.2-1 mcg/mL, +++ = 0.02-0.2 mcg/mL, ++++ = <0.02 mcg/mL
Red font indicates essentially no expectation of in vivo activity.
VIR-7831 (sotrovimab, Xevudy®) is GlaxoSmithKline’s IV MoAb infusion approved by EUA for 12 years and older for treating mild to moderate COVID-19 disease. Its distribution is controlled.
REGN 10933 (REGEN-COV, casirivimab) and REGN 10987 (REGEN-COV2, imdevimab) are Regeneron’s MoAb cocktail.
LY-CoV555’s (bamlanivimab, AbCellera®) EUA was revoked by the FDA at Lilly’s request in early 2021 as a single MoAb treatment. But it is part of a combination product with etesevimab (LY-CoV016) for use in those 12 years and older for treatment of moderate to severe COVID-19 disease or for prophylaxis in immune compromised persons.
AZD 7442 (Evushield) is the designation for the combination product that has AZD 8895 (tixagevimab) and AZD 1061 (cilgavimab); neither product is EUA approved for use alone. Can be used IV or IM. Protection is expected for six to 12 months.
References:
- Arora P, Sidarovich A, Krüger N, et al. B.1.617.2 enters and fuses lung cells with increased efficiency and evades antibodies induced by infection and vaccination. Cell Rep. 2021;37(2):109825. doi:10.1016/j.celrep.2021.109825
- Arora P, Kempf A, Nehlmeier I, et al. Delta variant (B.1.617.2) sublineages do not show increased neutralization resistance. Cell Mol Immunol. 2021;2557–2559. doi:10.1038/s41423-021-00772-y
- Dejnitrattisai W, Zhou D, Supasa P, et al. Antibody evasion by the P.1 strain of SARS-CoV-2. Cell. 2021;184(11):2939-2954.
- Rappazzo CG, Tse LV, Kaku, CI. et al. Broad and potent activity against SARS-like viruses by an engineered human monoclonal antibody. Science. 2021;371(6531):823-829.
- https://www.documentcloud.org/documents/21091950-centner-academy-response-dtd-10222021?responsive=1&title=1