Wide World of Vaccines
March 2022
Hopes and Concerns This Spring - COVID-19 Factoids; Mixed-Bag for RSV Prevention

Column Editor: Christopher Harrison, MD | Professor of Pediatrics, UMKC School of Medicine | Clinical Professor of Pediatrics, University of Kansas School of Medicine
COVID-19 prevalence concern. Globally, SARS-CoV-2 is showing signs of a new surge, causing new lockdowns in Asia (e.g., China/Hong Kong, New Zealand, South Korea) and rising worry that Europe removed too many restrictions too soon (e.g., the U.K., France, Italy) as the new BA.2 Omicron variant emerges. BA.2 now makes up 35% of total U.S. infections (Figure 1) and is increasing, but makes up <1% in Kansas and just over 3% in Missouri. BA.2 appears 40% more contagious than the prior BA.1 Omicron variant but does not seem to cause more severe disease, given current data. Wastewater surveillance has also shown an uptick in SARS-CoV-2 detections for the U.S. in the past three weeks. Such upticks usually herald a new surge in the U.S. As the U.S. is returning to more normal social interaction and less masking, the question is whether we will see a parallel rise in hospitalizations similar to that now reported in the U.K. just three weeks after they lifted restrictions. I hope not.
Pediatric Omicron disease concern. Omicron infections caused more pediatric hospitalizations in the past 3.5 months than in the 18 months previously, particularly in ≤6-month-olds. Figure 2.1 The good news is that most admissions were shorter (less than two days) than those due to Delta variant in the prior wave. Admissions were also shorter than respiratory syncytial virus (RSV) admissions during this past summer/fall RSV outbreak and appear mostly related to bronchiolitis or croup presentations.2 Let’s hope BA.2 doesn’t create even more pediatric disease and admissions.
SARS-CoV-2 vaccine hopeful news. Moderna indicated in a press release that they will soon be submitting their pediatric mRNA vaccine data. Moderna noted no concerns regarding safety; in addition, vaccine effectiveness (VE) was good in all age groups, unlike the <5-year-olds’ data from Pfizer. So, the Moderna pediatric vaccine could receive emergency use authorization (EUA) clearance sooner than the Pfizer vaccine for younger children, which now seems unlikely before summer. Also of note, Canada just approved the Moderna vaccine for 6- to 11-year-olds. On the other end of the age spectrum, Pfizer is asking for EUA for a second booster in persons ≥65 years old, likely to be given at four to nine months after the first booster. Not to be outdone, Moderna is asking the Food and Drug Administration for EUA for a second booster for all adults. I hope each vaccine request gets quick approval.
RSV Maternal Vaccination.
Concern: GSK paused their trial of immunizing pregnant women with a recombinant subunit pre-fusion RSV antigen (RSVPreF3) vaccine based on safety recommendations from an independent data safety monitoring board committee.3 The specific reason for pausing the trial was not given. However, in a phase 1-2 study in non-pregnant women, this same vaccine had shown “promising safety” per GSK. They reported that the vaccine had been well tolerated, and that it boosted pre-existing maternal RSV immunity at seven to 28 days post dose (14-fold increase in RSV-A and RSV-B neutralizing antibody titers). We can only wait for further clarity and hope the study can continue at some point.
More hope: Post hoc analysis of data from a randomized trial in 11 countries of a Novavax RSV fusion (F) protein nanoparticle vaccine to pregnant mothers revealed encouraging news for antibiotic stewardship despite not meeting the trial’s prespecified primary success criterion against proven RSV disease.4 Infants born to mothers randomized to the experimental RSV vaccine had a vaccine efficacy of 12.9% (95% CI: 1.3% to 23.1%) against being prescribed an antibiotic (VEPA) during their first three months of life compared to infants whose mothers got placebo. More specifically, VEPA for acute lower respiratory tract infections (LRTIs) was 16.9% (95% CI: 1.4% to 29.4%). Over the first three months of life, the effect/100 births was more notable in high-income countries than middle- to low-income countries (3.6 prescriptions given in high-income vs. 5.1 prescriptions in low- and middle-income countries). Among all antimicrobial prescribing prevented by maternal vaccination, 69% to 73% were for LRTI. Of note, a 71.3% (95% CI: 28.1% to 88.6%) reduction in prescriptions for acute otitis media (AOM) was also detected, but only in high-income countries. These data suggest that even though the vaccine was only partially effective in reducing proven RSV disease, it seems to have reduced acute respiratory infections to a degree that also led to less antibiotic use for LRTI and AOM.
RSV monoclonal antibody single dose for RSV prevention.
More hope: In a prior column 18 months ago, we discussed the significant protection provided to premature infants by one dose of an extended half-life monoclonal antibody (Mab) being developed jointly by AstraZeneca and Sanofi. The Mab (nirsevimab) targets the RSV pre-fusion protein. This month, new data were published on the nirsevimab’s effectiveness in preventing RSV infections in otherwise healthy term infants, enrolled at a median age of 2.6 months and representative of global pediatric diversity.5 Of the 1,490 randomized infants, 994 got nirsevimab and 496 got placebo. The Mab-efficacy was 74.5% (95% CI, 49.6% to 87.1%; P<0.001) for medically attended RSV infection and 62.1%; 95% CI, −8.6% to 86.8%; P = 0.07) for hospitalization. With all Mab, development of antibodies to the Mab that is a mouse/human hybrid molecule is a concern, but at near one year post-dosing, antidrug antibodies were detected in only 58/951 (6.1%) vs. 5/473 (1.1%) placebo recipients. Serious adverse events were reported in 6.8% Mab recipients and 7.3% placebo recipients.
Final hopes: With spring 2022 now upon us, we seem to be pivoting from the pandemic to more customary concerns and behavior. Let’s hope we are not jumping the gun (looking at you, July 2021) so that discussions of new approaches to RSV disease can be as important as discussions of COVID-19 related topics.
Figure 1. Proportions of current Omicron variants in the U.S.
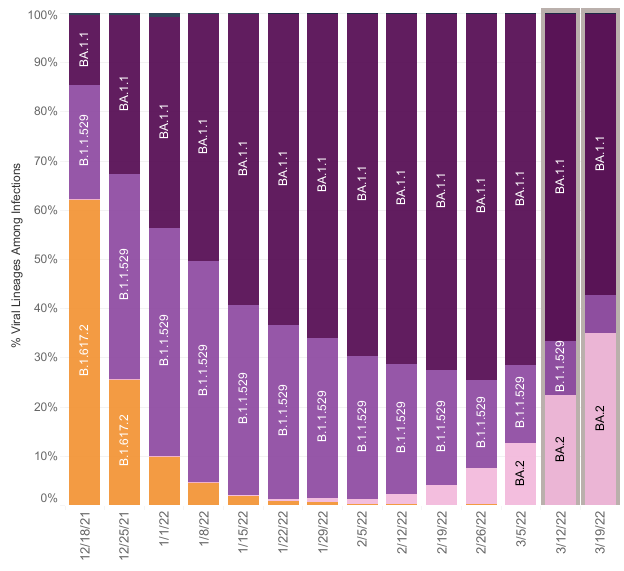
Source: CDC COVID-19 Data Tracker: Variant Proportions (accessed March 23, 2022)
Figure 2. Pediatric hospitalization in 2020 – 2022 in relation to Delta and Omicron waves.1
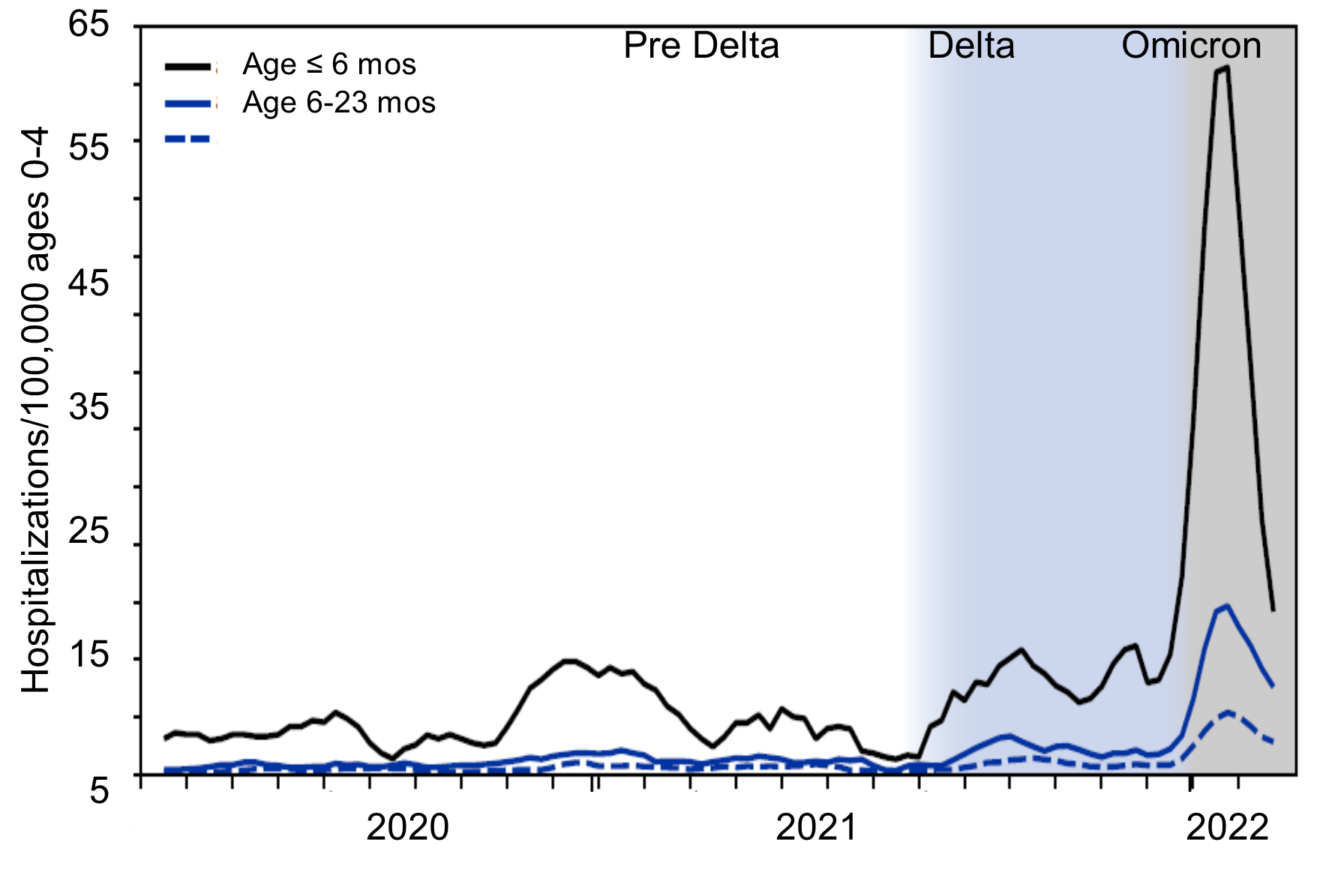
Source: Reference 1. All material in the MMWR series is in the public domain and may be used and reprinted without special permission.
References:
- Marks KJ, Whitaker M, Anglin O, et al; COVID-NET Surveillance Team. Hospitalizations of children and adolescents with laboratory-confirmed COVID-19 - COVID-NET, 14 States, July 2021-January 2022. MMWR Morb Mortal Wkly Rep. 2022;71(7):271-278.
- Meyer M, Ruebsteck E, Eifinger F, et al. Morbidity of respiratory syncytial virus-infections (RSV): RSV compared with SARS-CoV-2 infections in children aged 0 - 4 years in Cologne, Germany. J Infect Dis. 2022;jiac052. doi:10.1093/infdis/jiac052
- GSK pauses RSV maternal vaccine trial. https://www.reuters.com/business/healthcare-pharmaceuticals/gsk-pauses-late-stage-trial-rsv-vaccine-pregnant-women-2022-02-18/
- Lewnard JA, Fries LF, Cho I, Chen J, Laxminarayan R. Prevention of antimicrobial prescribing among infants following maternal vaccination against respiratory syncytial virus. Proc Natl Acad Sci U S A. 2022;119(12):e2112410119.
- Hammitt LL, Dagan R, Yuan Y, et al; MELODY Study Group. Nirsevimab for prevention of RSV in healthy late-preterm and term infants. N Engl J Med. 2022;386(9):837-846.