Wide World of Vaccines
February 2021
Like People, Almost All SARS-CoV-2 Viruses Are Genetically at Least a Bit Different – Does It Matter?

Author: Christopher Harrison, MD | Professor of Pediatrics, UMKC School of Medicine | Clinical Professor of Pediatrics, University of Kansas School of Medicine
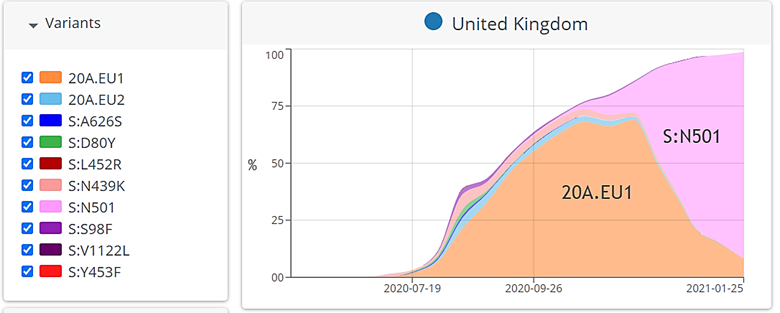
Variants in the News. “Variant” SARS-CoV-2 strains, particularly the United Kingdom B.1.1.7 variant is 40-60% more contagious than the prototype strain used to create vaccines because of S.N501 mutations.1,2 Figure 1, pink color. A South African (B.1.351) and a Brazilian (P.1) variant also are in the news with their E484K mutation. Most recently, the E484K mutation has been detected in addition to the S.N501 mutation in a dozen or so U.K. variants in England, seemingly double variants. All three newer variants are now in the U.S., the U.K. one being most widespread, but the U.K. double variant version so far is not in the U.S.
Variants that act differently raise concern that current vaccines may be less effective. So far, the two approved mRNA vaccines (Pfizer and Moderna) still protect against the U.K. strain. But current and impending vaccines (e.g., Astra Zeneca, Johnson & Johnson and Novavax) appear to provide variously lower potency in neutralizing the South African and Brazilian ones (45-80%) in vitro, and some in phase 2-3 studies (10% protection for the Astra Zeneca vaccine vs. the South African variant). RNA virus mutations leading to new strains emerging are not new; the original Chinese SARS-CoV-2 strain had already changed to a European variant (20A.EU1), Figure 1 orange color, before entering and dominating in the U.S. so far.
Expected mutation rate. Viruses give rise to mutants more frequently than do, for example, mammals, because viruses produce logarithmically more offspring than mammals. Each offspring has a chance to be a mutant, but each virus species can have different rates of mutation.3 Coronaviruses, for example, mutate approximately once out of every 3-6 million offspring, somewhat less frequent than influenza. But SARS-CoV-2 mutations are numerically more common because SARS-CoV-2 viral loads in patients’ secretions average 10-100 times higher than influenza virus; replication seems to last longer for SARS-CoV-2; and there are more SARS-CoV-2 infected people. Predictions estimate there will have been two to four times as many COVID-19 cases by summer compared to cases in an average influenza season. Together, these factors mean that at least one mutation in a SARS-CoV-2 copy likely arises in every person who gets infected. But is it as bad as it sounds?
Most mutations inconsequential. Most mutations are nonviable, meaning the mutant is crippled and cannot make more copies of itself. Of the viable mutations, most are so minor that they still do not change how the virus acts because the end-product, e.g., spike protein, still functions “normally.” These minor mutations, detectable by sequencing, rarely change contagion, virulence, response to antivirals or susceptibility to “natural” or vaccine-induced immunity. So far, almost all SARS-CoV-2 strains are still extremely close relatives classified into a small number of “clades.” Clades are virus groups with >90% identical nucleic acid sequences, and therefore clade members mostly act the same.
Consequential mutations. Mutants that act notably different usually have major or multiple concurrent nucleic acid mutations that change multiple amino acids and produce a major change in an important functional viral component. For example, the SARS-CoV-2 spike protein (target of the approved vaccines) has a changed structure in the Brazilian and South African variants. This means antibodies to the original strain’s spike protein no longer recognize the new spike protein’s shape as well, and so it may take up to double the less efficient antibody titer or even different antibodies to neutralize these variants. So, new variants may infect human cells even in current vaccine recipients or those with natural immunity from infection with older circulating strains, particularly if the antibody titer to the older strain is not high. Yet inefficient antibody still can protect against severe forms of disease, even if its protection against infection is incomplete (Johnson & Johnson vaccine vs. South African disease). So, less efficient antibody is better than none.
Scientists have sequenced nearly 400,000 SARS-CoV-2 isolates and still less than a dozen clades have arisen. Note: there are dozens of subclades4,5 ; but so far, each clade has sufficiently similar spike proteins that antibody induced by currently available vaccines or infection should work at least somewhat. The government-funded British universal sequencing effort showed its value when clinicians noted the recent explosion of new U.K. cases. Analysis of their sequence dataset showed the new variant, leading to new interventions. While the U.S. did not use massive national sequencing surveillance at first, we are catching up.2
Vaccine safety net. With new circulating strains partially evading current vaccines via spike protein mutations, new vaccines covering new variants are achievable in a few months using currently successful technology. Clinical testing/approvals will take a few more months. The variant vaccines could be given as solo injections in addition to current vaccines (similar to the initial H1N1 2019 influenza pandemic vaccines) or combined with a current vaccine into one multi-strain vaccine (like polio vaccines). Further, U.S. Army scientists have developed a vaccine that targets a “conserved” virus component other than the spike protein and will begin human trials soon, after success in animals.6 This could get around all the current spike protein variants’ resistance.
Bottom Line. Currently (mid-February 2021), two excellent vaccines remain effective against the two most common original (Chinese and European) viruses, but these and upcoming vaccines likely are less protective against the South African and Brazilian variants. Variant-specific new vaccines are now under development but are likely at least six months away from potential release.
Our role. Can we prevent more Godzilla-SARS-CoV-2 mutants from arising? The main preventive is reducing the trillions of viral replications that occur during rampant virus spread.4 We know how: by washing hands, social distancing, masks (double masking may be beneficial for those wearing masks that are not medical grade or have less than three layers), early diagnosis and contact tracing work. Preventing a single infection reduces the number of new virus copies by ~1 billion, also reducing chances of a new vaccine-evading variant. Preventing variant emergence is one more critical reason to get as many folks vaccinated as quickly as possible. Also, asymptomatic infections seem to be just as good of incubators for generating potential Godzilla-SARS-CoV-2 as symptomatic infections. So, it is critical to reduce all SARS-CoV-2 infections, even asymptomatic ones.
Figure 1. Variants in the U.K. Note rise of S:N501 (pink color) mutant starting in September.4
References:
- Rambaut A, Loman N, Pybus O, et al; COVID-19 Genomics Consortium U.K. Preliminary Genomic Characterisation of an Emergent SARS-CoV-2 Lineage in the U.K. Defined by a Novel Set of Spike Mutations. Virological.org. Posted Dec. 16, 2020. Accessed Jan. 14, 2021.
- Galloway SE, et al. Emergence of SARS-CoV-2 B.1.1.7 Lineage-United States, Dec. 29, 2020–Jan. 12, 2021. MMWR Early Release / Vol. 70 Jan. 15, 2021.
- Korber, et al., 2020, Cell 182, 812–827. Aug. 20, 2020 https://doi.org/10.1016/j.cell.2020.06.043.
- Hodcraft EB. CoVariants: SARS-CoV-2 Mutations and Variants of Interest. https://github.com/hodcroftlab/covariants/blob/master/README.md.
- Lauring AS, Hodcroft EB. Genetic Variants of SARS-CoV-2—What Do They Mean? Pp. E1-4. jama.com (Reprinted) JAMA Published online Jan. 6, 2021.
- Army’s COVID-19 Vaccine May Hold Key to Beating Coronavirus Mutations. Reported in McClatchyDC.com.