What's the Diagnosis?
September 2022
Visual Diagnosis
Column Author and Editor: Joe Julian, MD, MPHTM, FAAP | Hospitalist, Internal Medicine – Pediatrics | Clinical Associate Professor, Internal Medicine and Pediatrics, UMKC School of Medicine
A 3-year-old male is directly admitted to the hospitalist service for evaluation and management of an elevated lead level. Patient initially had a lead level of 58 approximately six weeks ago. The family was contacted by the health department to be reevaluated, and they saw their physician approximately two weeks ago for repeat testing and evaluation. The family was just contacted about the results (128 mcg/dL), and the patient was referred for direct admission.
Patient does not have lethargy, constipation, abdominal pain, pica or behavioral changes. He has ongoing speech delay. No home assessment has been done. The home was built in 1900 and has peeling paint inside. Recently, other homes in the neighborhood have been demolished. The family drinks city water. They have no occupations or hobbies associated with lead exposure.
Vital signs and examination are within age-appropriate limits. Neurologic examination is notable only for speech delay, as noted above, but no encephalopathy.
Pertinent labs & imaging
- Repeat lead level results at 103 mcg/dL (critical level >/= 45)
- Total bilirubin is mildly elevated at 1.6 mg/dL (1.2 upper limit of normal) with 0.2 mg/dL direct
- Hemoglobin is low at 10 g/dL with a normal MCV of 78 fL
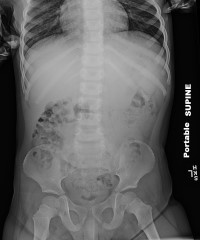
An abdominal X-ray is obtained and shown below:
Question
Which of the following is the best initial step for therapy?
- Perform whole bowel irrigation with polyethylene glycol (PEG)
- Administer oral succimer and intravenous edetate calcium disodium (CaEDTA)
- Perform endotracheal intubation with elevated intracranial pressure precautions
- Administer oral D-penicillamine
Answer
B. Administer oral succimer and intravenous edetate calcium disodium (CaEDTA)
There is no “safe” lead level. Even low blood lead levels (BLLs) (even those <5 mcg/dL) can have cognitive and behavioral effects. Children are more susceptible than adults to lead toxicity as they have a higher absorption of lead in the gastrointestinal tract, often have concurrent iron deficiency, which increases lead absorption, and may not have a completely formed blood-brain barrier. The goal is appropriate screening and initiation of timely therapy when indicated. The choice of chelation therapy is dependent upon the blood lead level and the presence (or absence) of encephalopathy and seizures.
Children with a BLL 70 or higher should have therapy initiated in the hospital with the assistance of a toxicologist. This 3-year-old patient has a severely elevated lead level but fortunately does not show evidence of encephalopathy or elevated intracranial pressure, which would change his immediate management. Of the above choices, initiation of oral succimer and intravenous CaEDTA would be the most appropriate. If this patient had encephalopathy, substitution of intramuscular dimercaprol for oral succimer would be appropriate. The CaEDTA would be given intramuscularly as well.
Whole bowel irrigation is not appropriate at this time. There are no “lead flecks” appreciated on the abdominal radiograph. Additionally, iron is absorbed in the small intestine (not the large), so this treatment method would be useful only if there were obvious sources of lead in the small intestine. At best, this treatment has mixed evidence of being successful. There is a moderate stool burden shown in the radiograph, but constipation is a clinical diagnosis and does not always correlate with radiographic findings (and is also not the patient’s primary medical issue).
Oral D-penicillamine is essentially a non-first-line agent at this point due to its side effect profile and the availability of better therapies. Its use is often considered if there are allergic or resource availability issues with other treatments.
Case Resolution
The patient was admitted to the hospital for four days with toxicology consultation and received both oral succimer and intravenous CaEDTA. His lead level trended down to 53 mcg/dL at discharge. He was prescribed oral succimer for an additional 14 days. Patient’s family was instructed to have a repeat level drawn at two and four weeks after completion of succimer. However, these lead levels were not obtained.
The patient unfortunately had an elevated lead level of 59 mcg/dL approximately six weeks after completion of succimer therapy. It was postulated that he likely had ongoing exposure that contributed to the elevation in levels (especially given rapid improvement with second round of chelation therapy) rather than mobilization from bone. He was discharged to the home of a family member who did not have any concerns with environmental lead and completed an additional two weeks of oral succimer.
Additional Points of Consideration
- Patients with a detectable blood lead level need appropriate follow-up given lead’s impact on development (new reference level is 3.5 mcg/dL).
- An environmental evaluation should be completed to determine source of lead exposure.
- People exposed to the environmental source should be screened for lead toxicity (especially siblings).
- Chelation therapy is not recommended until a blood lead level of 45 mcg/dL is reached as there are no differences in outcomes at lower levels.
Clinician Resources
American Academy of Pediatrics – Lead Exposure
Centers for Disease Control – Childhood Lead Poisoning Prevention
