Outbreaks, Alerts and Hot Topics
July 2021
Respiratory Viruses in the Middle of Summer and New Updates from ACIP

Column Editor: Angela Myers, MD, MPH | Director, Division of Infectious Diseases | Professor of Pediatrics, UMKC School of Medicine | Medical Editor, The Link Newsletter
Across the U.S. we have seen a rise in respiratory viruses over the past month. This is unusual timing since we are generally past respiratory virus season at this point in the year. However, with mask requirements being lifted and more people participating in social activities, we have seen a rise in circulating respiratory viral pathogens. The Centers for Disease Control and Prevention (CDC) released a Health Advisory through the Health Alert Network (HAN)1 on June 10, recommending broader testing for respiratory syncytial virus (RSV) in the setting of negative COVID-19 testing. Many southern states have seen an especially high increase in cases in Health and Human Services regions 4 (Alabama, Florida, Georgia, Kentucky, Mississippi, North Carolina, South Carolina, Tennessee) and 6 (Arkansas, Louisiana, New Mexico, Oklahoma, Texas).
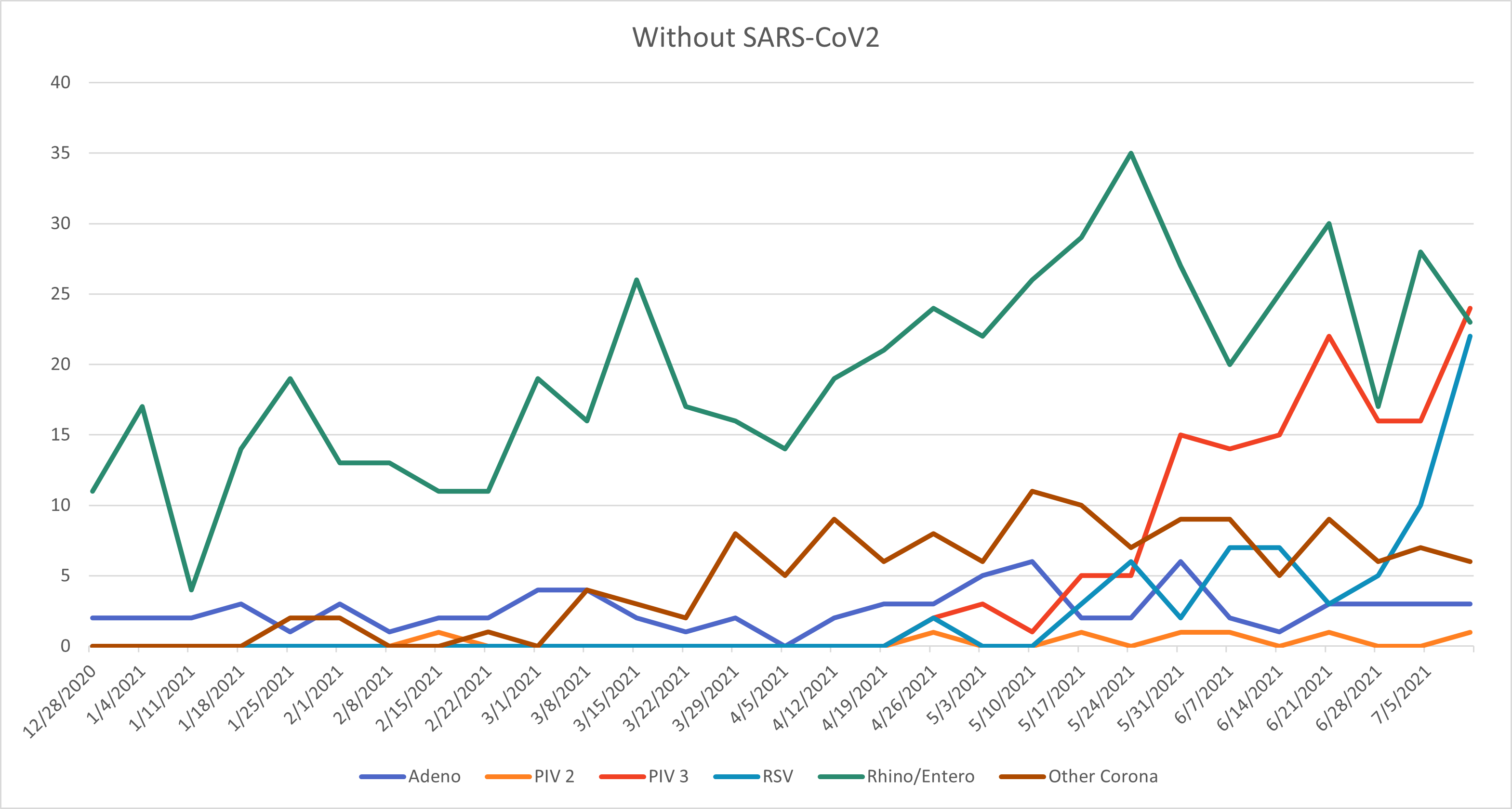
The increase in cases has led to re-initiation of palivizumab in some states including Florida, Louisiana and Texas. The table shows the percent of positive cases over the last five weeks in Kansas and Missouri, with Florida as a comparison. We are clearly seeing an uptick in both Kansas and Missouri, although not as high as in states like Florida. Over the last few weeks at Children’s Mercy, we have been seeing some RSV, but we have also found adenovirus, rhinovirus/enterovirus (remember that the PCR platform cannot differentiate between these viruses), parainfluenza and seasonal coronavirus in addition to SARS-CoV-2. Figures 1a and 1b show the last six months of respiratory virus data at Children’s Mercy with and without SARS-CoV-2 included. Low-level activity was seen throughout for rhinovirus/enterovirus and adenovirus (this is typical), no influenza last winter (very atypical), and more recently an increase in parainfluenza (mostly type 3 which is typical in the spring), some RSV, and seasonal coronavirus, while SARS-CoV-2 has declined.
Why is this happening? If 2020 has taught us anything, it is that unusual things can and will happen. It makes sense that cases of SARS-CoV-2 have declined as more people have become vaccinated, although there are areas of increasing spread and we are seeing an increase in cases as the Delta variant is quickly becoming the dominant strain in our states and across the country. It also makes sense that we are seeing these respiratory viruses re-emerge as more people are going without masks and are out and about.
Additionally, we have a cohort of children and seniors who hadn’t been exposed to these seasonal viruses for over a year (since March 2020) and thus are at higher risk for severe infection. Each year pre-COVID-19 pandemic, RSV deaths in the U.S. occurred in 100-500 children <5 years of age and in 14,000 adults ≥65 years, with well over 230,000 hospitalizations among these two age groups. Nearly everyone would agree that 2020-21 has been strange to say the least and appears to be continuing that way. The state of Missouri has recommended restarting palivizumab for children at high risk for severe RSV infection in our community. No word from Kansas yet, but stay tuned. Children’s Mercy pharmacists are currently in discussion with the manufacturer of palivizumab and insurance companies to mitigate barriers to provide palivizumab while RSV rates remain elevated in Kansas and Missouri.
Advisory Committee on Immunization Practices (ACIP) Updates:
When the ACIP met near the end of June they made several updated recommendations including allowing co-administration of influenza vaccine with COVID-19 vaccine, acknowledgement that the 2021-22 season will be the first one in which all available influenza vaccines are quadrivalent, and the Flucelvax (virus grown in cultured cells rather than hen eggs) made by Seqirus was approved for children as young as 2 years (decrease from 4 years) in March of this year and will now be available to this age group.2 Additionally, the ACIP is now recommending two doses of rabies vaccine seven days apart for pre-exposure prophylaxis in children instead of three doses. This updated recommendation was based on data from 190 children who had adequate antibody titers following a two-dose series of rabies vaccine. A booster dose is recommended for those with ongoing exposure risk within three years of the initial series. While rabies pre-exposure prophylaxis is not often used, it is recommended in instances of international travel to remote areas that do not have ready access to medical care, including rabies vaccine or rabies immune globulin. Finally, dengue vaccine is now recommended for children 9-16 years of age who live in endemic areas (e.g., Puerto Rico) and have had previous dengue infection. This vaccine has been shown to have an 82% efficacy in preventing symptomatic dengue infection.
Table. Percent of positive RSV cases by state per week since end of May 2021.
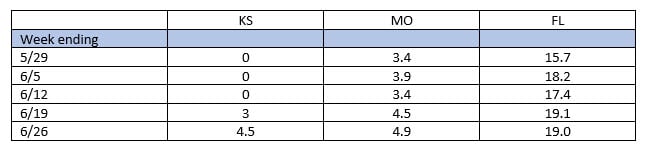
Figure 1a. Number of respiratory viruses, including SARS-CoV-2, identified December 2020 through June 2021.
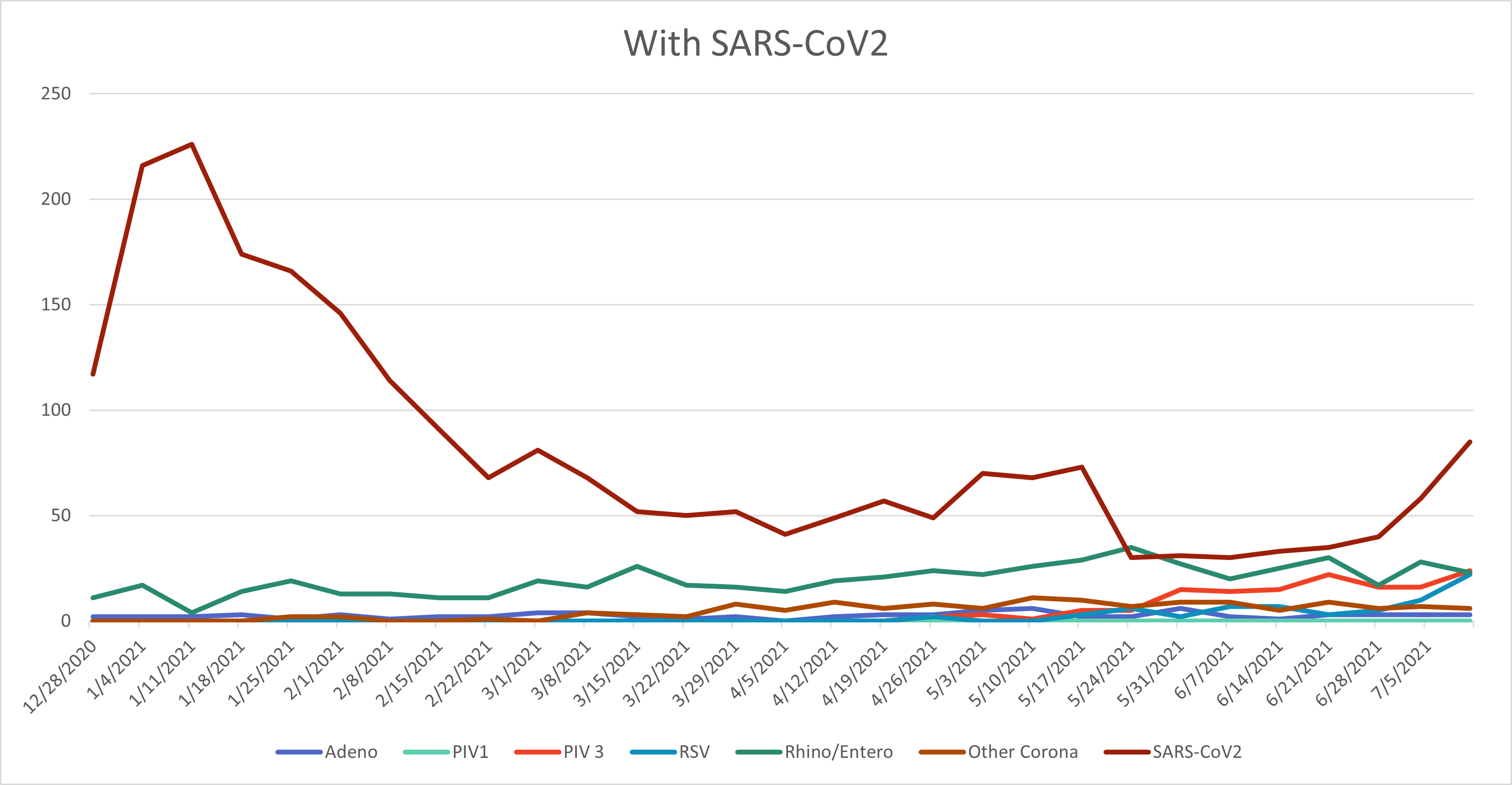
Figure 1b. Number of respiratory viruses, without SARS-CoV-2, identified December 2020 through June 2021.
References:
- Accessed June 30, 2021. https://emergency.cdc.gov/han/2021/han00443.asp.
- Accessed June 30, 2021. https://www.aappublications.org/news/2021/06/24/acip-flu-rabies-dengue-062421.