Academic Curriculum
Pediatric Clinical Pharmacology Fellowship
Overview
The research fellowship program in Pediatric Clinical Pharmacology at Children's Mercy Kansas City provides didactic education in a variety of clinical pharmacology topics and hands on training in translational and clinical research.
The objectives for each fellow are to: 
Core didactics
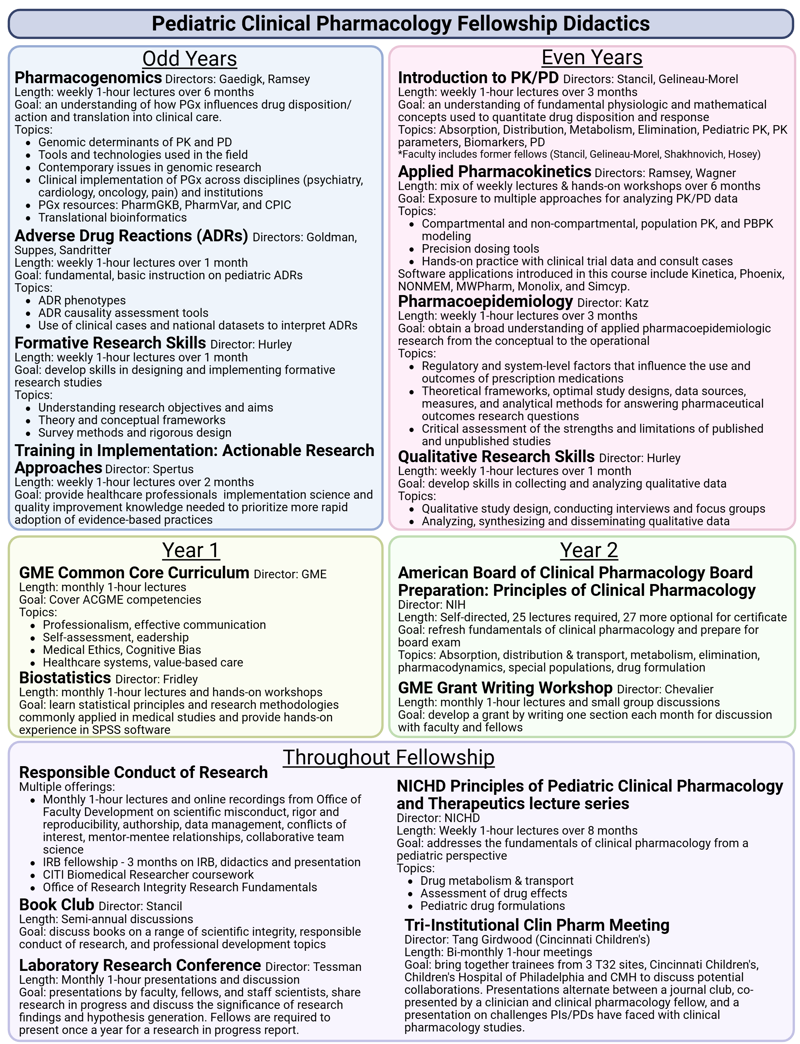
Other local instruction

View other GME or hospital conferences.
Advanced degree offerings
Master of Science. All trainees entering the program who do not have a graduate degree (MS or PhD) in a biomedical science have the option to enroll in one of two Master’s programs at KUMC or UMKC. It is anticipated that trainees will spend 2 to 2.5 years completing degree requirements. This extended period may be required to accommodate the schedule for the required core didactic clinical pharmacology offerings from the program at Children's Mercy and the external rotations.
Elective rotations

Our Pediatric Clinical Pharmacology Fellowship leadership team
Laura Ramsey, PhD
Co-Program Director
lramsey@cmh.edu
Jonathan Wagner, DO
Co-Program Director
jbwagner@cmh.edu
Stephani Stancil, PhD, APRN
Associate Program Director
slstancil@cmh.edu
Rachel Laws, MBA, C-TAGME
Program Coordinator
rmlaws@cmh.edu